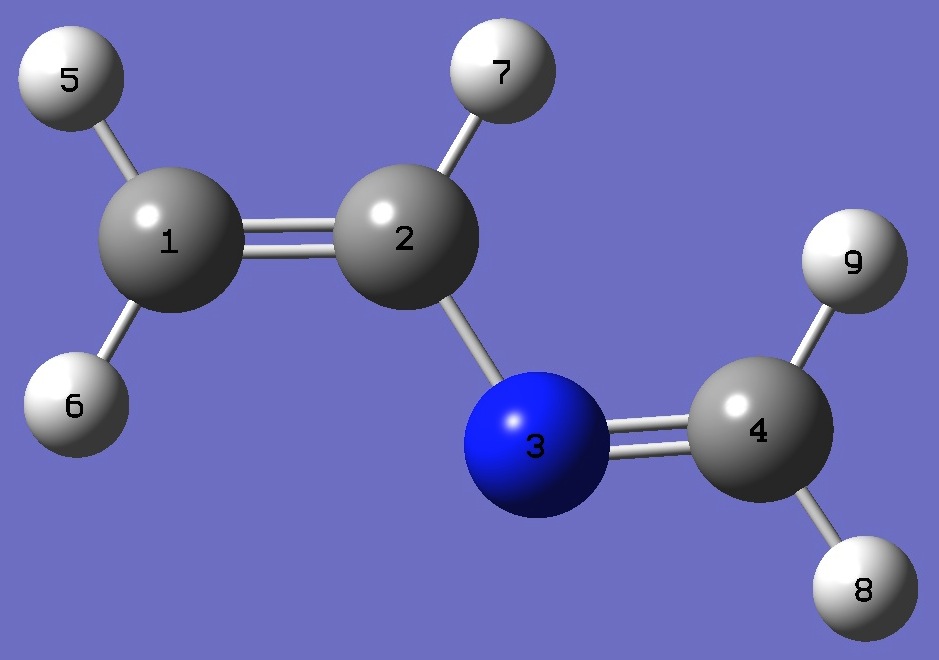
| Table 1. 14N nqcc's in 2-Azabutadiene (MHz). Calculation was made on (1) B3LYP/6-311+G(3df,3pd) and (2) B3LYP/aug-cc-pVTZ optimized structures. |
||||||||
| Calc (1) |
Calc (2) | Expt [1] | ||||||
| Xaa |
0.970 |
0.970 |
0.5(10) |
|||||
| Xbb | - |
4.792 |
- |
4.791 |
- |
4.4(11) |
||
| Xcc | 3.822 |
3.821 |
3.9(5) |
|||||
| |Xab| |
1.254 |
1.254 |
||||||
| RMS | 0.36 (12. %) |
0.36 (12. %) | ||||||
| RSD | 0.030 (1.3 %) | 0.030 (1.3 %) | ||||||
| Xxx | 1.231 |
1.231 |
||||||
| Xyy | 3.822 | 3.821 |
||||||
| Xzz | - |
5.054 |
- |
4.052 |
||||
| ETA | 0.513 |
0.512 |
||||||
| Øz,a | 78.24 |
78.24 |
||||||
| Øa,bi * | ||||||||
| Øz,bi | ||||||||
| |
||||||||
| Table 3. 2-Azabutadiene: Rotational constants (MHz) and dipole moments (D): ropt(1) = B3LYP/6-311+G(3df,3pd) and ropt(2) = B3LYP/aug-cc-pVTZ optimized structure. | ||||
| ropt(1) | ropt(2) | Expt [1] | ||
| A | 48657. |
48630. |
47186.010(23) |
|
| B | 4892. |
4891. |
4886.5325(27) |
|
| C | 4445. |
4444. |
4430.0673(23) |
|
| |µa| |
0.40 |
0.40 |
0.443(33) |
|
| |µb| | 1.50 |
1.50 |
1.896(74) |
|
| Table 4. 2-Azabutadiene: Quartic Centrifugal Distortion Constants (MHz). Calc = B3LYP/cc-pVTZ. |
||||
| Calc | Expt [1] |
|||
| Delta_J |
0.000955 |
0.000995(11) |
||
| Delta_JK |
- 0.00804 |
- 0.00801(29) |
||
| Delta_K |
0.337 |
0.3406(32) |
||
| delta_j |
0.000117 |
0.00011743(46) |
||
| delta_k |
0.00472 |
0.005158(51) |
||