|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
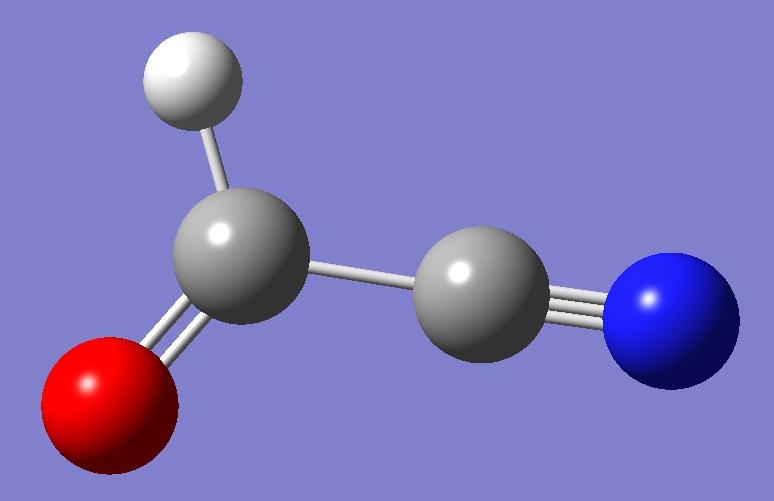
HCOCN
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Nitrogen
|
|
|
Nuclear
Quadrupole Coupling Constants |
|
|
|
in Formyl Cyanide |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Calculation was made of the nitrogen
nqcc's in formyl cyanide on an approximate equilibrium structure, ~re derived by MP2/aug-cc-pVTZ(G03) optimization with empirically corrected bond lengths. These calculated nqcc's are compared with the experimental
values [1] in Table 1. Structure parameters are given in Table 2. |
|
|
|
|
|
|
|
|
|
|
|
|
In Table 1, subscripts a,b,c refer to the principal axes of the inertia
tensor, subscripts x,y,z to the principal axes of the nqcc tensor.
The nqcc y-axis is chosen coincident with the inertia c-axis, these
are perpendicular to the plane of the molecule. Ø (degrees)
is the angle between its subscripted parameters. ETA = (Xxx
- Xyy)/Xzz. |
|
|
RMS is the root mean square
difference between calculated and experimental nqcc's (percentage of
average experimental nqcc). RSD is the residual stand deviation
of calibration of the B3PW91/6-311+G(df,pd) model for calculation of
the nqcc's. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
Table 1. Nitrogen nqcc's
in HCOCN (MHz). Calculation was made on ~re structure. |
|
| |
|
|
|
|
|
|
|
|
|
|
|
Calc. |
|
Expt. [1] |
|
| |
|
|
|
|
|
|
|
|
14N |
Xaa |
- |
4.415 |
- |
4.330(19) |
|
|
|
Xbb |
|
1.410 |
|
1.531(14) |
|
|
|
Xcc |
|
3.005 |
|
2.799(29) |
|
|
|
|Xab| |
|
1.529 |
|
|
|
|
|
|
|
|
|
|
|
|
|
RMS |
|
0.147 (5.1 %) |
|
|
|
|
|
RSD |
|
0.030 (1.3 %) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Xxx |
|
1.787 |
|
|
|
|
|
Xyy |
|
3.005 |
|
|
|
|
|
Xzz |
- |
4.792 |
|
|
|
|
|
ETA |
|
0.254 |
|
|
|
|
|
Øz,a |
|
13.85 |
|
|
|
|
|
Øa,CN |
|
13.36 |
|
|
|
|
|
Øz,CN |
|
0.49 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Table 2. Molecular structure
parameters (Å and degrees). |
|
|
|
|
|
|
|
|
|
CH |
|
1.0962 |
|
|
|
CO |
|
1.2044 |
|
|
|
CC |
|
1.4693 |
|
|
|
CN |
|
1.1574 |
|
|
|
HCO |
|
123.08 |
|
|
|
HCC |
|
114.83 |
|
|
|
CCN |
|
178.62 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
[1] M.Bogey, C.Demuynck, J.L.Destombes, Yo.Vallee,
and J.L.Ripoll, J.Mol.Spectrosc. 172,344(1995). |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH3COCN |
HCOCl |
CH3COCl |
CH3CDO |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table of Contents |
|
|
|
|
|
Molecules/Nitrogen |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
HCOCN.html |
|
|
|
|
|
|
Last
Modified 29 Dec 2008 |
|
|
|
|
|
|
|
|
|
|
