|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NH2-C(=O)-CH3
|
|
PDF
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Nitrogen |
|
|
|
Nuclear
Quadrupole Coupling Constants |
|
|
in Acetamide
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Calculation of the
nitrogen nqcc's in acetamide was made here on the molecular
structures given by HF/6-311++G(3df,3pd) and HF/aug-cc-pVTZ optimizations. These are
compared with the
experimental nqcc's [1] in Tables 1 and 2. Structure parameters are
given in Table 3, rotational constants in Table 4.
|
|
|
|
|
|
|
|
|
|
|
|
|
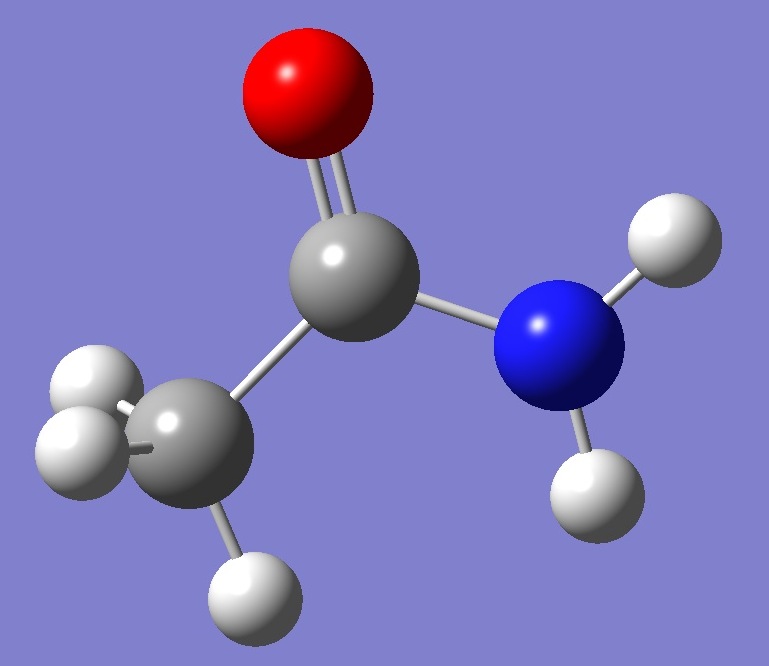
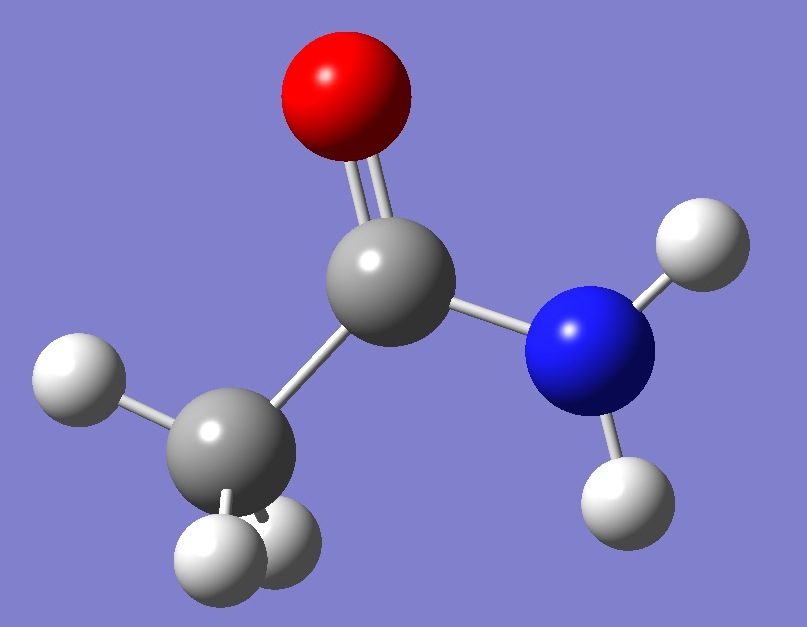
Within the constraint of Cs symmetry, which is here assumed, the in-plane methyl hydrogen Hs is either trans or cis with respect to the oxygen atom, as shown below: |
|
|
|
|
|
|
|
|
|
|
|
|
|
anti-Acetamide |
|
syn-Acetamide |
|
|
|
|
|
|
|
|
|
|
|
|
|
 |
 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Energy: At the HF/6-311++G(3df,3pd) and HF/aug-cc-pVTZ(G03) levels of theory, Esyn < Eanti by 0.12 and 0.044 kcal/mol, respectively. However, Eanti < Esyn by 0.21 kcal/mol at the B3PW91/6-311+G(df,pd) level of theory for
both HF/6-311++G(3df,3pd) and HF/aug-cc-pVTZ optimized structures.
Calculation was made of the nqcc's on both conformers. Agreement between
calculated and experimental nqcc's is much better for nqcc's calculated
on anti-acetamide (Table 1) than for those calculated on syn-acetamide
(Table 2). |
|
|
|
|
|
|
|
|
|
|
|
|
In Tables 1 and 2, subscripts a,b,c refer to the
principal axes of the inertia tensor; x,y,z to the principal axes
of the nqcc tensor.
Ø (degrees) is the angle between its subscripted
parameters. ETA = (Xxx - Xyy)/Xzz. |
|
|
|
|
|
|
|
|
|
|
|
|
RMS is the root mean square
difference between calculated and experimental diagonal nqcc's (percentage of the
average of the magnitudes of the experimental nqcc's). RSD is the
calibration residual standard deviation of the B3PW91/6-311+G(df,pd) model for calculation of nitrogen nqcc's. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
Table 1. 14N nqcc's in anti-Acetamide (MHz). Calculation was made
on the (1) HF/6-311++G(3df,3pd) and (2) HF/aug-cc-pVTZ(G03) optimized structures. |
|
| |
|
|
|
|
|
|
|
|
|
|
|
Calc. (1)
|
|
Calc. (2) |
|
Expt. [1] |
|
| |
|
|
|
|
|
|
|
|
|
Xaa |
|
1.952 |
|
1.950 |
|
1.9368(24) |
|
|
Xbb |
|
2.019 |
|
2.019 |
|
2.0090(22) |
|
|
Xcc |
- |
3.971 |
- |
3.968 |
- |
3.9458(22) |
|
|
|Xab| |
|
0.048 |
|
0.051 |
|
0.0998(78) |
|
|
|
|
|
|
|
|
|
|
|
RMS |
|
0.018 (0.69 %) |
0.016 (0.61 %) |
|
|
|
RSD |
|
0.030 (1.3 %) |
0.030 (1.3 %) |
|
|
|
|
|
|
|
|
|
|
|
|
Xxx |
|
1.927 |
|
1.923 |
|
1.876(6) |
|
|
Xyy |
|
2.044 |
|
2.046 |
|
2.098(6) |
|
|
Xzz |
- |
3.971 |
- |
3.968 |
- |
3.9458(22) |
|
|
ETA |
|
0.019 |
|
0.031 |
|
|
|
|
Øy,a |
|
117.53 |
|
117.89 |
|
|
|
|
Øa,NC |
|
133.00 |
|
132.83 |
|
|
|
|
Øy,NC |
|
15.46 |
|
14.93 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
Table 2. 14N nqcc's in syn-Acetamide (MHz). Calculation was made
on the (1) HF/6-311++G(3df,3pd) and (2) HF/aug-cc-pVTZ(G03) optimized structures. |
|
| |
|
|
|
|
|
|
|
|
|
|
|
Calc. (1)
|
|
Calc. (2) |
|
Expt. [1] |
|
| |
|
|
|
|
|
|
|
|
|
Xaa |
|
2.023 |
|
2.021 |
|
1.9368(24) |
|
|
Xbb |
|
2.092 |
|
2.092 |
|
2.0090(22) |
|
|
Xcc |
- |
4.115 |
- |
4.113 |
- |
3.9458(22) |
|
|
|Xab| |
|
0.030 |
|
0.033 |
|
0.0998(78) |
|
|
|
|
|
|
|
|
|
|
|
RMS |
|
0.112 (4.2 %) |
0.118 (4.5 %) |
|
|
|
RSD |
|
0.030 (1.3 %) |
0.030 (1.3 %) |
|
|
|
|
|
|
|
|
|
|
|
|
Xxx |
|
2.012 |
|
2.008 |
|
1.876(6) |
|
|
Xyy |
|
2.103 |
|
2.105 |
|
2.098(6) |
|
|
Xzz |
- |
4.115 |
- |
4.113 |
- |
3.9458(22) |
|
|
ETA |
|
0.019 |
|
0.024 |
|
|
|
|
Øy,NC |
|
18.68 |
|
17.55 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
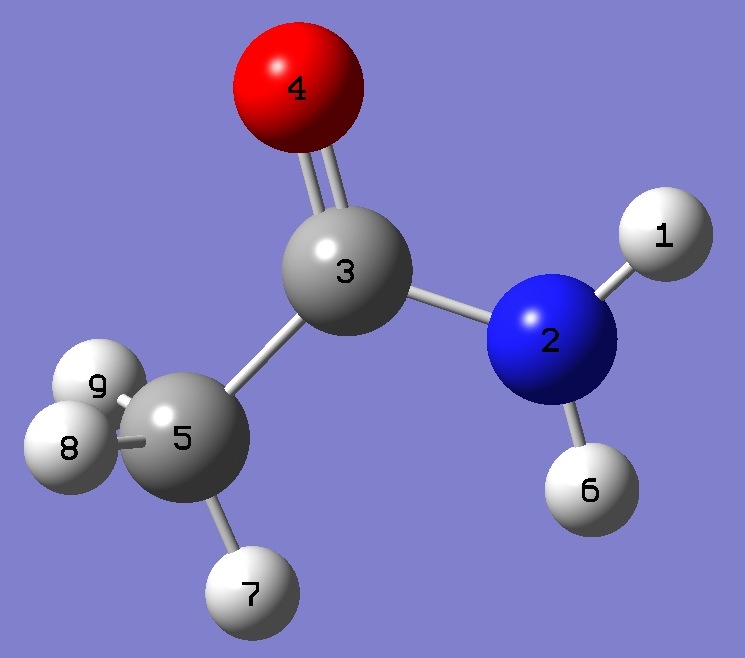
| Table 3. anti-Acetamide. Molecular structure parameters (Å
and degrees). |
| |
|
|
|
|
ropt (1) = HF/6-311++G(3df,3pd) optimization. |
|
ropt (2) = HF/aug-cc-pVTZ(G03) optimization. |
| |
|
|
|
| Point Group: Cs |
|
ropt (1) |
ropt (2) |
|
|
|
|
|
NH(1) |
0.9914 |
0.9912 |
| NH(6) |
0.9879 |
0.9876 |
| NC |
1.3513 |
1.3516 |
| C=O |
1.1924 |
1.1940 |
| CC |
1.5112 |
1.5112 |
| CH(7) |
1.0812 |
1.0815 |
| CH(8) |
1.0811 |
1.0814 |
| CH(9) |
1.0811 |
1.0814 |
|
CNH(1) |
118.48 |
118.50 |
|
CNH(6) |
119.17 |
119.17 |
| Dihedral angles? |
NC=O |
122.14 |
122.13 |
| See Z-Matrix. |
NCC |
116.22 |
116.20 |
|
CCH(7) |
113.47 |
113.44 |
| syn-Acetamide? Z-Matrix. |
CCH(8) |
108.30 |
108.32 |
|
CCH(9) |
108.30 |
108.32 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
| Table 4. anti-Acetamide. Rotational Constants (MHz). Normal Species. |
| |
|
|
|
|
|
ropt (1) = HF/6-311++G(3df,3pd) optimization. |
|
ropt (2) = HF/aug-cc-pVTZ(G03) optimization. |
| |
|
|
|
|
|
|
Calc. ropt (1) |
Calc. ropt (2) |
Expt. [1] |
|
|
|
|
|
|
A |
11231.5 |
11212.5 |
10839.43(27) |
|
B |
9367.9 |
9366.0 |
9285.20(12) |
|
C |
5271.8 |
5267.1 |
5156.15(11) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
[1] N.Heineking and H.Dreizler, Z.Naturforsch. 48a,787(1993).
|
|
|
|
|
|
|
|
|
|
|
|
|
Related ... |
|
|
|
|
|
|
"Ground and first excited torsional
states of acetamide" V.V.Hyushin, E.A.Alekseev, S.F.Dyubko,
I.Kleiner, and J.T.Hougen, J.Mol.Spectrosc. 227,115(2004). |
|
|
"Reinvestigation of the Microwave
Spectrum of Acetamide" R.D.Suenram, G.Yu.Golubiatnikov, I.I.Leonov,
J.T.Hougen, J.Ortigoso, L.Kleiner, and G.T.Fraser, J.Mol.Spectrosc.
208,188(2001). |
|
|
"Acetamide,
a challenge to theory and experiment? ... " S.Samdal, J.Mol.Struct.
440,165(1998). "It is not likely that the syn conformation ... is the
stable conformation in the gaseous state."
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Formamide |
N-Methylacetamide |
N-Ethylformamide |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table of Contents |
|
|
|
|
|
Molecules/Nitrogen |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Acetamide.html |
|
|
|
|
|
|
Last
Modified 25 Dec 2005 |
|
|
|
|
|
|
|
|
|
|


