|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH3COCl |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Chlorine |
|
|
|
Nuclear
Quadrupole Coupling Constants |
|
|
|
in
Acetyl Chloride |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Chlorine nqcc's in acetyl chloride
were determined by Hayashi et al. [1], which investigators also derived
a substitution molecular structure. Calculation of the nqcc's was
made here on this structure and on a ~re structure given by MP2/aug-cc-pVTZ with empirically corrected C-C,
C=O, and CCl bond lengths. Calculated and experimental nqcc's
are compared in Table 1, structure parameters in Table 2. |
|
|
|
|
|
|
|
|
|
|
|
|
In Table 1, RMS is the root mean
square difference between calculated and experimental diagonal nqcc's
(percentage of the average of the magnitudes of the experimental
nqcc's). RSD is the calibration residual standard deviation for
the B1LYP/TZV(3df,2p) model for calculation of the chlorine
nqcc's. |
|
|
|
|
|
|
|
|
|
|
|
|
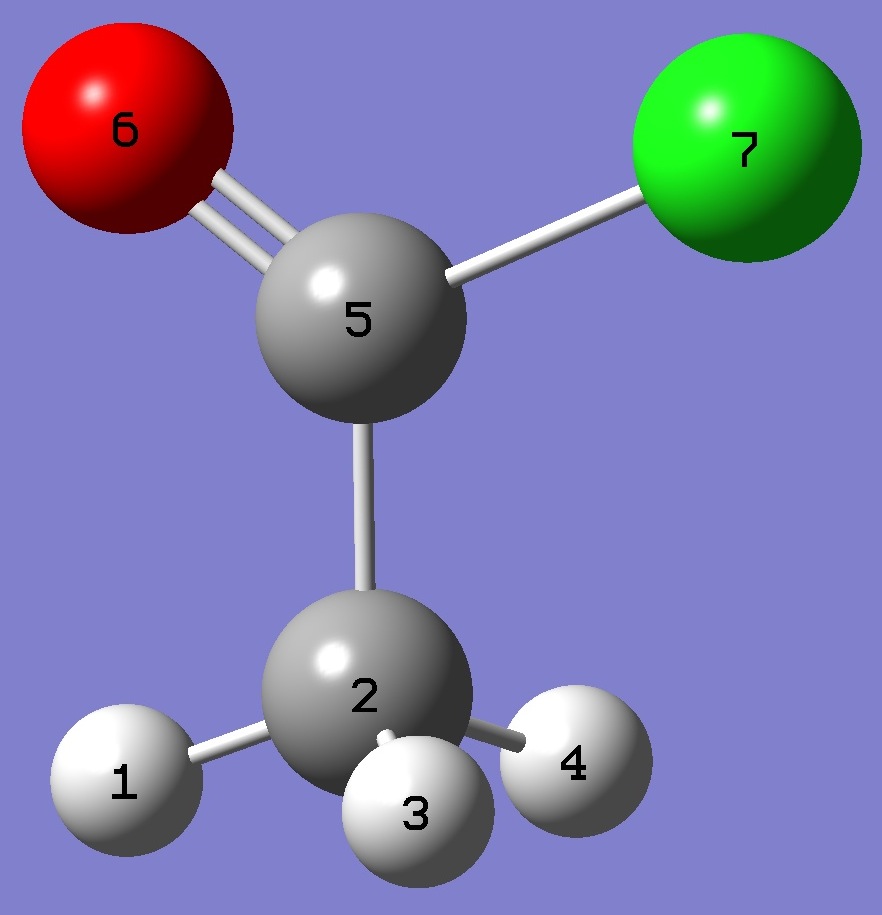
Subscripts a,b,c refer to the
principal axes of the inertia tensor; x,y,z to the principal axes
of the nqcc tensor. The nqcc y-axis is chosen coincident with the
inertia c-axis, these are perpendicular to the molecular symmetry
plane.
Ø (degrees) is the angle between its subscripted
parameters. ETA = (Xxx - Xyy)/Xzz. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
Table 1. Chlorine
nqcc's in CH3COCl (MHz). Calculation was made on
the rs and ~re structures. |
|
| |
|
|
|
|
|
|
|
|
|
|
|
Calc / rs |
|
Calc / ~re |
|
Expt. [1] |
|
| |
|
|
|
|
|
|
|
|
|
Xaa (35Cl) |
- |
60.68 |
- |
59.10 |
- |
58.437(11) |
|
|
Xbb |
|
38.73 |
|
37.56 |
|
36.493(11) |
|
|
Xcc |
|
21.95 |
|
21.54 |
|
21.944(11) |
|
|
|Xab| |
|
8.80 |
|
9.70 |
|
|
|
|
|
|
|
|
|
|
|
|
|
RMS |
|
1.83 (4.7 %) |
|
0.76 (2.0 %) |
|
|
|
|
RSD |
|
0.49 (1.1 %) |
|
0.49 (1.1 %) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Xxx |
|
39.51 |
|
38.52 |
|
|
|
|
Xyy |
|
21.95 |
|
21.54 |
|
|
|
|
Xzz |
- |
61.46 |
- |
60.07 |
|
|
|
|
ETA |
- |
0.286 |
- |
0.283 |
|
|
|
|
Øz,a |
|
5.02 |
|
5.68 |
|
|
|
|
Øa,CCl |
|
5.97 |
|
6.06 |
|
|
|
|
Øz,CCl |
|
0.95 |
|
0.38 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Xaa(37Cl) |
- |
47.87 |
- |
46.62 |
- |
45.701(16) |
|
|
Xbb |
|
30.57 |
|
29.64 |
|
28.530(16) |
|
|
Xcc |
|
17.30 |
|
16.98 |
|
17.172(16) |
|
|
|Xab| |
|
6.70 |
|
7.46 |
|
|
|
|
|
|
|
|
|
|
|
|
|
RMS |
|
1.72 (5.6 %) |
|
0.84 (2.8 %) |
|
|
|
|
RSD |
|
0.44 (1.1 %) |
|
0.44 (1.1 %) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Table 2. Acetyl Chloride.
Molecular structure parameters, rs [1] and ~re
(Å and degrees) |
| |
|
|
|
|
 |
|
rs |
~re |
|
|
|
| CC |
1.485(6) |
1.4939 |
| C=O |
1.209(6) |
1.1828 |
| CCl |
1.783(2) |
1.7971 |
| CH(1) |
1.080(1) |
1.0858 |
| CH(3,4) |
1.090(1) |
1.0883 |
| CCO |
126.92(130) |
128.08 |
| CCCl |
113.62(55) |
111.42 |
| CCH(1) |
109.94(38) |
109.39 |
|
|
CCH(3,4) |
108.88(40) |
109.06 |
|
|
H(1)CH(3,4) |
110.85(15) |
110.81 |
|
|
H(3)CH(4) |
107.35(23) |
107.67 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
[1] M.Hayashi, N.Inada, and
Y.Niide, J.Mol.Struct. 352/353,325(1995). |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
O=CHCl |
O=CFCl |
O=CCl2 |
|
|
|
S=CCl2 |
S=CFCl |
s-t-CH2CHCOCl |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table of Contents |
|
|
|
|
|
Molecules/Chlorine |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH3COCl.html |
|
|
|
|
|
|
Last
Modified 11 Aug 2009 |
|
|
|
|
|
|
|
|
|
|

