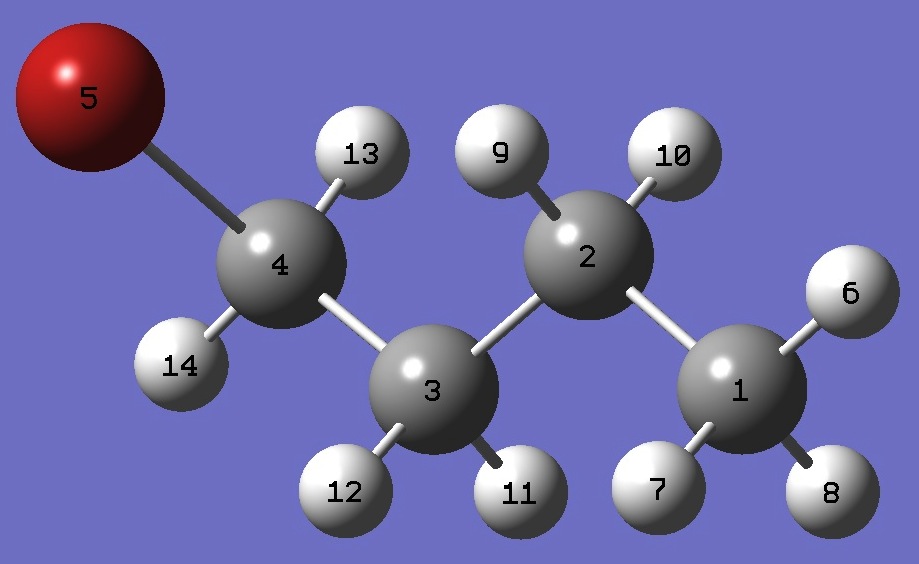
| |
||||||||
| Table 1. 79Br nqcc's in 1-Bromobutane, GA (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(3df,3pd) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C and CBr bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | 86.47 |
87.68 |
87.17(10) |
|||||
| Xbb | 167.75 |
166.92 |
166.53(6) * |
|||||
| Xcc | - |
254.22 |
- |
254.60 |
- |
253.70(6) * |
||
| |Xab| ** |
396.63 |
397.26 |
394.70(44) |
|||||
| |Xac| | 72.31 |
72.25 |
72.26(236) |
|||||
| |Xbc| | 87.68 |
87.38 |
87.38(18) |
|||||
| RMS | 0.86 (0.62 %) |
0.70 (0.42 %) |
||||||
| RSD | 1.58 (0.39 %) |
1.58 (0.39 %) | ||||||
| Xxx | - |
265.94 |
- |
266.25 |
- |
264.89(197) |
||
| Xyy | - |
276.07 | - |
276.37 | - |
274.83(154) |
||
| Xzz | 542.01 |
542.62 |
539.71(63) |
|||||
| ETA | 0.0187 |
0.0187 |
||||||
| Øz,CBr | 0.68 |
0.69 |
||||||
| |
||||||||
| Table 2. 81Br nqcc's in 1-Bromobutane, GA (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(3df,3pd) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C and CBr bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | 73.29 |
74.30 |
73.83(93) |
|||||
| Xbb | 139.17 |
138.48 |
138.18(47) * |
|||||
| Xcc | - |
212.46 |
- |
212.78 |
- |
212.01(47) * |
||
| |Xab| ** | 331.51 |
332.02 |
329.92(43) |
|||||
| |Xac| |
60.37 |
60.32 |
59.81(271) |
|||||
| |Xbc| |
72.99 |
72.74 |
74.04(149) |
|||||
| RMS | 0.70 (0.49 %) |
0.55 (0.39 %) |
||||||
| RSD | 1.38 (0.40 %) |
1.38 (0.40 %) | ||||||