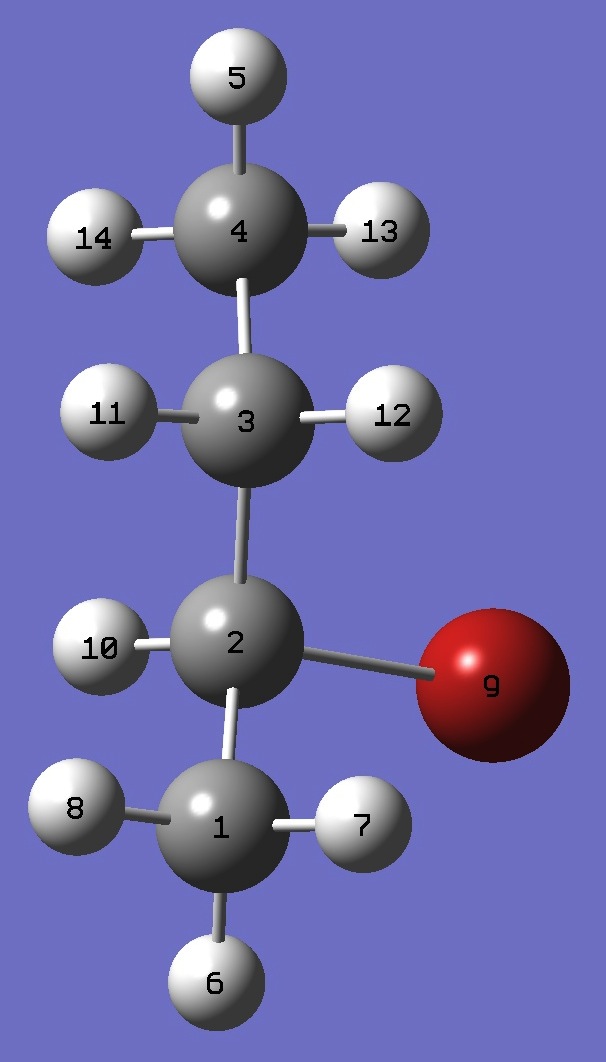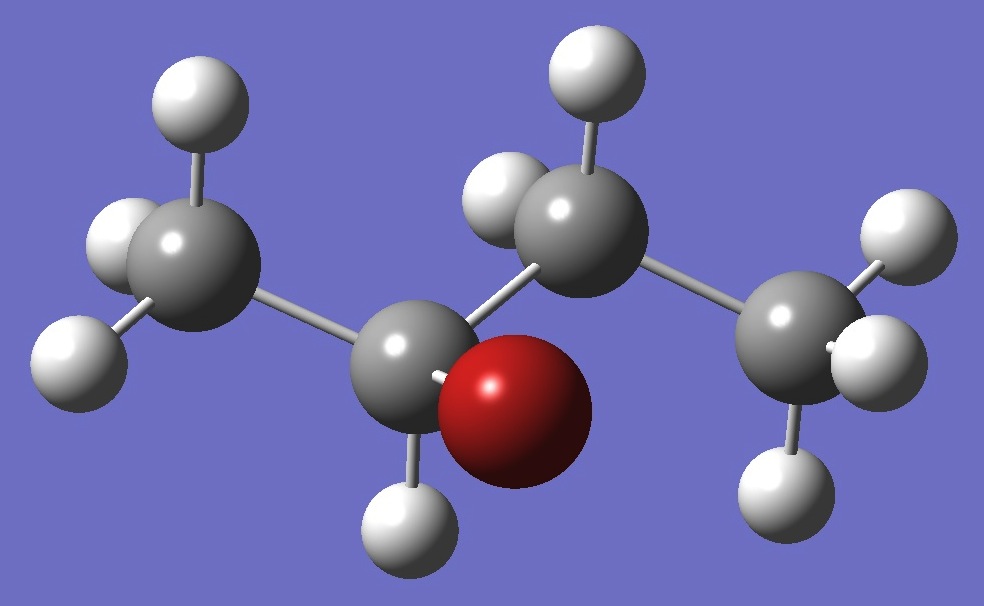
| |
||||||||
| Table 1. 79Br nqcc's in 2-Bromobutane, G+ (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(3df,3pd) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C and CBr bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | 304.09 |
306.93 |
300.1367(2) |
|||||
| Xbb | - 83.07 |
- 85.69 |
- 82.2620 * |
|||||
| Xcc | - |
221.01 |
- |
221.24 |
- |
217.8747 * |
||
| Xab | - |
324.64 |
- |
323.35 |
321.18(6) |
|||
| Xac | - |
141.75 |
- |
141.93 |
- |
140.93(2) |
||
| Xbc | 82.43 |
81.80 |
- 82.16(1) |
|||||
| RMS | 2.95 (1.47 %) |
4.81 (2.40 %) |
||||||
| RSD | 1.58 (0.39 %) |
1.58 (0.39 %) | ||||||
| Xxx | - |
256.82 | - |
256.95 | ||||
| Xyy | - |
267.72 | - |
267.90 | ||||
| Xzz | 524.54 |
524.86 |
||||||
| ETA | 0.0208 |
0.0209 |
||||||
| Øz,CBr | ||||||||
| |
||||||||
| Table 2. 81Br nqcc's in 2-Bromobutane, G+ (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(3df,3pd) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected CBr bond length. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | 257.21 |
258.54 |
253.8573(2) |
|||||
| Xbb | - 72.50 |
- 74.63 |
- 71.7862 * |
|||||
| Xcc | - |
184.72 |
- |
184.91 |
- |
182.0711 * |
||
| Xab | - |
269.38 |
- |
268.30 |
266.64(9) |
|||
| Xac | - |
118.69 |
- |
118.83 |
- |
117.00(2) |
||
| Xbc | 68.12 |
67.59 |
- 68.25(2) |
|||||
| RMS | 2.50 (1.48 %) |
4.02 (2.37 %) |
||||||
| RSD | 1.38 (0.40 %) |
1.38 (0.40 %) | ||||||