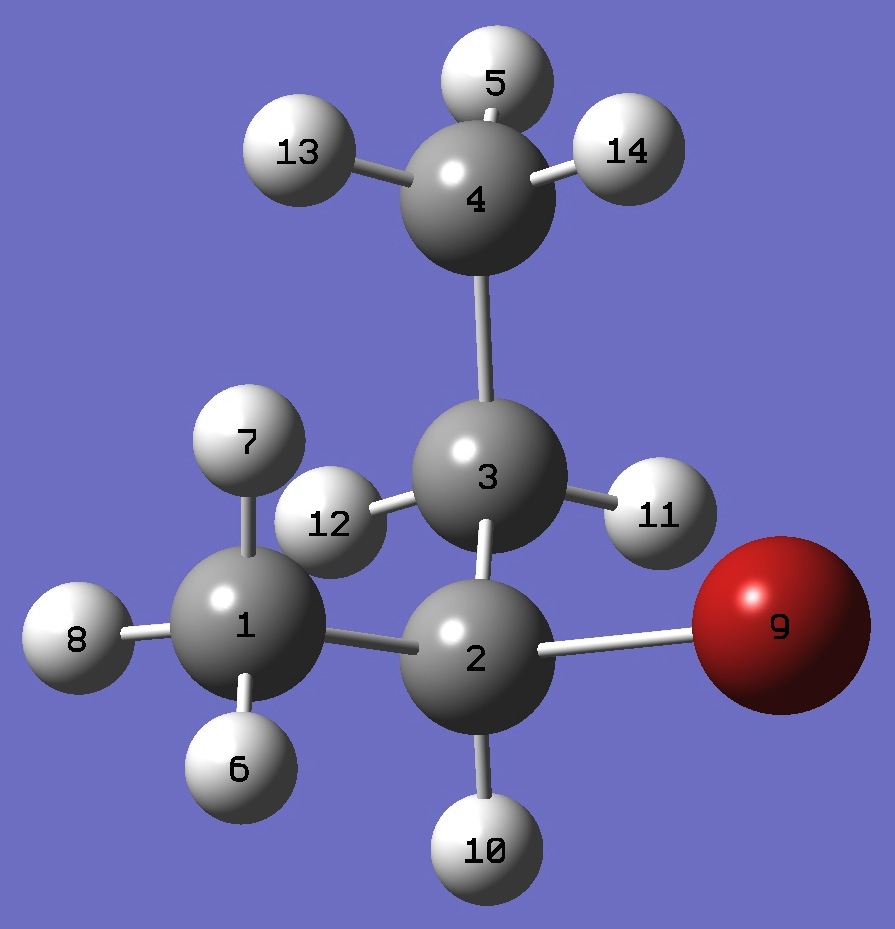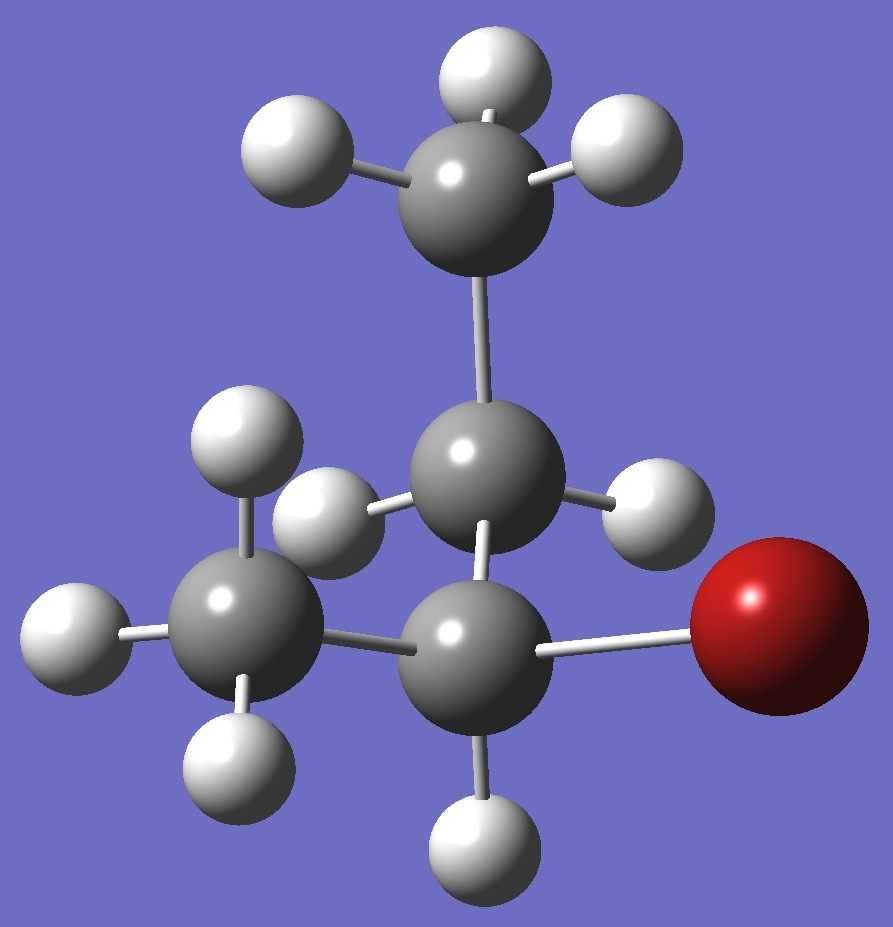
| |
||||||||
| Table 1. 79Br nqcc's in 2-Bromobutane, G- (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(3df,3pd) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C and CBr bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | 324.40 |
325.94 |
317.134(1) |
|||||
| Xbb | - |
132.28 |
- |
133.34 |
- |
127.125(2) * |
||
| Xcc | - |
192.12 |
- |
192.59 |
- |
190.009(2) * |
||
| Xab | - |
275.72 |
- |
275.13 |
- |
274.8(6) |
||
| Xac | - |
204.12 |
- |
203.90 |
200.6(9) |
|||
| Xbc | 96.54 |
95.95 |
- 96.21(9) |
|||||
| RMS | 5.28 (2.50 %) |
6.34 (3.03 %) |
||||||
| RSD | 1.58 (0.39 %) |
1.58 (0.39 %) | ||||||
| Xxx | - |
261.78 | - |
261.92 | ||||
| Xyy | - |
263.36 | - |
263.48 | ||||
| Xzz | 525.14 |
525.40 |
||||||
| ETA | 0.0030 |
0.0030 |
||||||
| Øz,CBr | ||||||||
| |
||||||||
| Table 2. 81Br nqcc's in 2-Bromobutane, G- (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(3df,3pd) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected CBr bond length. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | 272.21 |
273.48 |
266.182(1) |
|||||
| Xbb | - |
111.53 |
- |
112.41 |
- |
107.283(2) * |
||
| Xcc | - |
160.68 |
- |
161.07 |
- |
158.899(2) * |
||
| Xab | - |
229.55 |
- |
229.06 |
- |
230.8(7) |
||
| Xac | - |
170.51 |
- |
170.32 |
165.2(1) |
|||
| Xbc | 80.16 |
79.67 |
- 78.70(2) |
|||||
| RMS | 4.38 (2.47 %) |
5.30 (2.99 %) |
||||||
| RSD | 1.38 (0.40 %) |
1.38 (0.40 %) | ||||||