| |
||||||||
| Table 1. 127I nqcc's in CH3C(=O)I (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(d,p) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C, C=O, and CH bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | - | 1586.5 | - | 1570.5 | - | 1563(2) | ||
| Xbb | 929.3 | 919.7 | 914(2) |
|||||
| Xcc | 657.2 |
650.8 | 649(3) * |
|||||
| Xab | - 135.6 |
- 132.2 |
- 135.6(2) |
|||||
| RMS | 16.8 (1.62 %) | 5.5 (0.53 %) | ||||||
| RSD | 15.2 (1.23 %) | 15.2 (1.23 %) | ||||||
| Xxx | 936.6 | 926.7 | ||||||
| Xyy | 657.2 | 650.8 | ||||||
| Xzz | - | 1593.8 | - | 1577.5 | ||||
| ETA | - | 0.175 | - | 0.175 | ||||
| Øz,a | 3.08 | 3.03 | ||||||
| Øa,CI | 3.49 | 3.52 | ||||||
| Øz,CI | 0.41 | 0.49 | ||||||
| |
||||||||
| Table 2. 127I nqcc's in CH3C(=O)I (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(2d,2p) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C, C=O, and CH bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | - | 1583.5 | - | 1567.2 | - | 1563(2) | ||
| Xbb | 937.4 | 927.3 | 914(2) |
|||||
| Xcc | 646.1 |
639.9 | 649(3) |
|||||
| Xab | - 140.4 |
- 138.4 |
- 135.6(2) |
|||||
| RMS | 18.0 (1.73 %) | 9.9 (0.95 %) | ||||||
| RSD | 15.2 (1.23 %) | 15.2 (1.23 %) | ||||||
| Xxx | 945.2 | 935.0 | ||||||
| Xyy | 646.1 | 639.9 | ||||||
| Xzz | - | 1591.3 | - | 1574.9 | ||||
| ETA | - | 0.188 | - | 0.187 | ||||
| Øz,a | 3.18 | 3.17 | ||||||
| Øa,CI | 3.50 | 3.61 | ||||||
| Øz,CI | 0.32 | 0.44 | ||||||
| |
||||||||
| Table 3. 127I nqcc's in CH3C(=O)I (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(3d,3p) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C, C=O, and CH bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | - | 1582.4 | - | 1568.4 | - | 1563(2) | ||
| Xbb | 930.5 | 921.9 | 914(2) |
|||||
| Xcc | 651.9 |
646.5 | 649(3) |
|||||
| Xab | - 137.0 |
- 135.0 |
- 135.6(2) |
|||||
| RMS | 14.8 (1.42 %) | 5.7 (0.55 %) | ||||||
| RSD | 15.2 (1.23 %) | 15.2 (1.23 %) | ||||||
| Xxx | 938.0 | 929.2 | ||||||
| Xyy | 651.9 | 646.5 | ||||||
| Xzz | - | 1589.9 | - | 1575.7 | ||||
| ETA | - | 0.180 | - | 0.179 | ||||
| Øz,a | 3.11 | 3.09 | ||||||
| Øa,CI | 3.47 | 3.54 | ||||||
| Øz,CI | 0.35 | 0.45 | ||||||
| |
||||||||
| Table 4. 127I nqcc's in CD3C(=O)I (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(d,p) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C, C=O, and CH bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | - | 1565.5 | - | 1550.1 | - | 1541(3) | ||
| Xbb | 908.3 | 899.4 |
892(2) |
|||||
| Xcc | 657.2 |
650.7 | 649(4) |
|||||
| Xab | - 265.9 |
- 260.3 |
- 258.5(10) |
|||||
| RMS | 17.6 (1.72 %) | 6.9 (0.67 %) | ||||||
| RSD | 15.2 (1.23 %) | 15.2 (1.23 %) | ||||||
| |
||||||||
| Table 5. 127I nqcc's in CD3C(=O)I (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(2d,2p) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C, C=O, and CH bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | - | 1561.7 | - | 1545.8 | - | 1541(3) | ||
| Xbb | 915.6 | 905.8 |
892(2) |
|||||
| Xcc | 646.1 |
639.9 | 649(4) |
|||||
| Xab | - 272.6 |
- 268.7 |
- 258.5(10) |
|||||
| RMS | 18.2 (1.77 %) | 9.9 (0.97 %) | ||||||
| RSD | 15.2 (1.23 %) | 15.2 (1.23 %) | ||||||
| |
||||||||
| Table 6. 127I nqcc's in CD3C(=O)I (MHz). | ||||||||
| Calc (1) was made on the MP2/6-311+G(3d,3p) optimized molecular structure. | ||||||||
| Calc (2) was made on this same structure but with empirically corrected C-C, C=O, and CH bond lengths. | ||||||||
| Calc (1) | Calc (2) | Expt. [1] | ||||||
| |
||||||||
| Xaa | - | 1561.2 | - | 1547.6 | - | 1541(3) | ||
| Xbb | 909.3 | 901.1 |
892(2) |
|||||
| Xcc | 651.9 |
646.5 | 649(4) |
|||||
| Xab | - 267.6 |
- 263.9 |
- 258.5(10) |
|||||
| RMS | 15.5 (1.51 %) | 6.6 (0.64 %) | ||||||
| RSD | 15.2 (1.23 %) | 15.2 (1.23 %) | ||||||
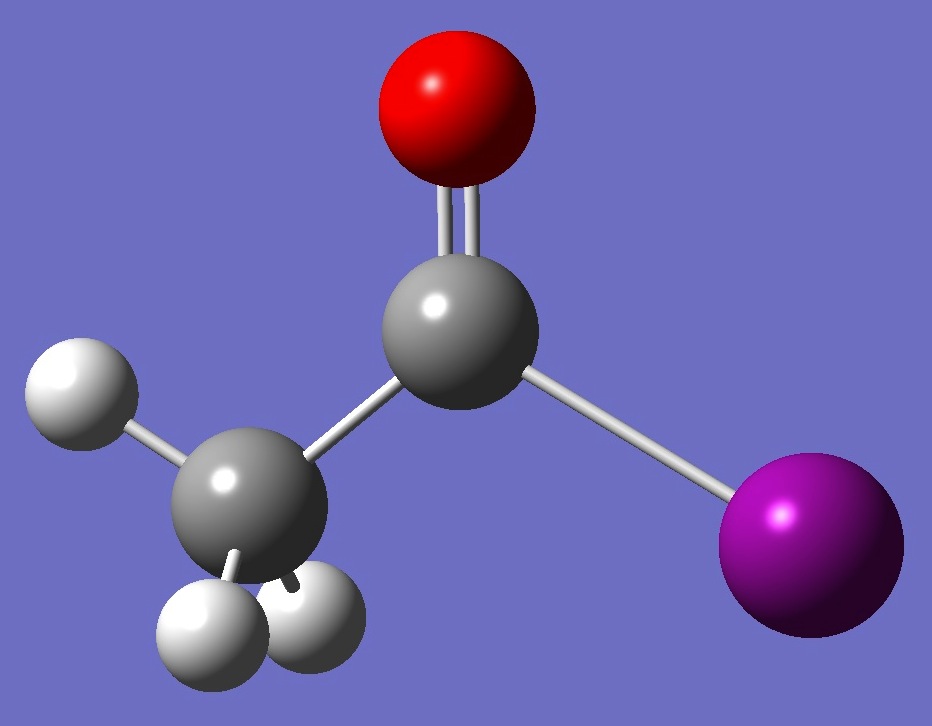
| Table 7.
Acetl Iodide. Molecular structure
parameters (Å and
degrees). Corrected bond lengths are given in parentheses. |
|||
| H C,1,B1 H,2,B2,1,A1 H,2,B3,1,A2,3,D1,0 C,2,B4,1,A3,3,D2,0 O,5,B5,2,A4,1,D3,0 I,5,B6,2,A5,1,D4,0 |
|||
| MP2/6-311+G(d,p) |
|||
| B1=1.09193128 (1.0889) B2=1.09220835 (1.0893) B3=1.09220835 (1.0893) B4=1.50385811 (1.4974) B5=1.18687532 (1.1781) B6=2.24269435 A1=111.02564887 A2=111.02564887 A3=109.15022941 A4=128.28750444 A5=111.48333766 D1=120.06005045 D2=-119.96997477 D3=0. D4=180. |
|||
| MP2/6-311+G(2d,2p) | |||
| B1=1.08497532 (1.0889) B2=1.08604823 (1.0893) B3=1.08604823 (1.0893) B4=1.49903188 (1.4974) B5=1.18870062 (1.1781) B6=2.20432012 A1=110.88440346 A2=110.88440346 A3=109.43861989 A4=127.31968145 A5=111.97927556 D1=119.43985928 D2=-120.28007036 D3=0. D4=180. |
|||
| MP2/6-311+G(3d,3p) | |||
| B1=1.08662869 (1.0889) B2=1.08760316 (1.0893) B3=1.08760316 (1.0893) B4=1.49974819 (1.4974) B5=1.18683553 (1.1781) B6=2.22896324 A1=111.03643981 A2=111.03643981 A3=109.32558666 A4=128.05942989 A5=111.53997809 D1=120.01132409 D2=-119.99433795 D3=0. D4=180. |
|||