|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
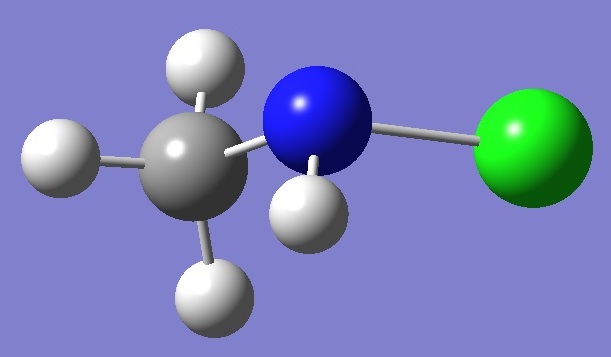
CH3NHCl |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Chlorine and Nitrogen
|
|
|
Nuclear
Quadrupole Coupling Constants |
|
|
in N-Chloro-methylamine
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cl and N nqcc's in CH3NHCl, as well as all monodeuterated-methyl species, were determined by Caminati et al. [1.2]. |
|
|
|
|
|
|
|
|
|
|
|
|
Calculation of the chlorine and nitrogen nqcc tensors was made here on an ropt structure given by MP2/aug-cc-pVTZ(G03) optimization. These
are compared with the experimental nqcc's in Tables 1 -3.
Structure parameters are compared with those of reference [2]
in
Table 4; rotational constants are given in
Table 5. |
|
|
|
|
|
|
|
|
|
|
|
|
In Tables 1 - 3, subscripts a,b,c refer to the principal axes of the inertia
tensor, subscripts x,y,z to the principal axes of the nqcc tensor. Ø (degrees)
is the angle between its subscripted parameters. ETA = (Xxx
- Xyy)/Xzz. |
|
|
RMS is the root mean square
difference between calculated and experimental nqcc's (percentage of
the average experimental nqcc). RSD is the residual standard deviation
of calibration of the model for calculation of
the nqcc's. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
Table 1. Cl and N nqcc's in CH3NH35Cl (MHz). Calculation was made on the MP2/aug-cc-pVTZ(G03) optimized structure, ropt. |
|
| |
|
|
|
|
|
|
|
|
|
|
|
Calc. |
|
Expt. [1] |
|
| |
|
|
|
|
|
|
|
|
35Cl |
Xaa |
- |
66.82 |
- |
67.02(6) |
|
|
|
Xbb |
|
18.78 |
|
18.90(7) |
|
|
|
Xcc |
|
48.04 |
|
48.12 |
|
|
|
|Xab|
|
|
56.55 * |
|
|
|
|
|
|Xac|
|
|
6.09 |
|
|
|
|
|
|Xbc|
|
|
1.55 |
|
|
|
|
|
|
|
|
|
|
|
|
|
RMS |
|
0.14 (0.32 %) |
|
|
|
|
|
RSD |
|
0.49 (1.1 %) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Xxx |
|
46.11 |
|
|
|
|
|
Xyy |
|
49.09 |
|
|
|
|
|
Xzz |
- |
95.20 |
|
|
|
|
|
ETA |
|
0.031 |
|
|
|
|
|
Øz,NCl |
|
0.65 |
|
|
|
|
|
|
|
|
|
|
|
|
14N |
Xaa |
|
4.856 |
|
4.63(9) |
|
|
|
Xbb |
- |
0.038 |
- |
0.20(5) |
|
|
|
Xcc |
- |
4.818 |
- |
4.43 |
|
|
|
|Xab| |
|
1.874 * |
|
|
|
|
|
|Xac| |
|
0.201 |
|
|
|
|
|
|Xbc| |
|
2.823 |
|
|
|
|
|
|
|
|
|
|
|
|
|
RMS |
|
0.27 (8.9 %) |
|
|
|
|
|
RSD |
|
0.030 (1.3 %) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Xxx |
|
0.668 |
|
|
|
|
|
Xyy |
|
5.548 |
|
|
|
|
|
Xzz |
- |
6.216 |
|
|
|
|
|
ETA |
|
0.785 |
|
|
|
|
|
Øz,NCl |
|
80.88 |
|
|
|
|
|
Øz,NH |
|
60.06 |
|
|
|
|
|
Øz,NC |
|
64.27 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
* The algebraic sign of the product XabXacXbc is negative. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
Table 2. Cl and N nqcc's in CH3NH37Cl (MHz). Calculation was made on the MP2/aug-cc-pVTZ(G03) optimized structure, ropt. |
|
| |
|
|
|
|
|
|
|
|
|
|
|
Calc. |
|
Expt. [1] |
|
| |
|
|
|
|
|
|
|
|
37Cl |
Xaa |
- |
52.93 |
- |
53.7(2) |
|
|
|
Xbb |
|
15.07 |
|
16.0(2) |
|
|
|
Xcc |
|
37.86 |
|
37.7 |
|
|
|
|Xab| |
|
44.36 * |
|
|
|
|
|
|Xac| |
|
4.79 |
|
|
|
|
|
|Xbc| |
|
1.20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
RMS |
|
0.7 (2.0 %) |
|
|
|
|
|
RSD |
|
0.44 (1.1 %) |
|
|
|
|
|
|
|
|
|
|
|
|
14N |
Xaa |
|
4.868 |
|
5.1(8) |
|
|
|
Xbb |
- |
0.050 |
|
|
|
|
|
Xcc |
- |
4.818 |
|
|
|
|
|
|Xab| |
|
1.859 |
|
|
|
|
|
|Xac| |
|
0.191 |
|
|
|
|
|
|Xbc| |
|
2.825 |
|
|
|
|
|
|
|
|
|
|
|
|
|
RSD |
|
0.030 (1.3 %) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
* The algebraic sign of the product XabXacXbc is negative. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
Table 3. Cl and N nqcc's in CH2DNH35Cl (MHz). Calculation was made on the MP2/aug-cc-pVTZ(G03) optimized structure, ropt. See below for numbering of D atoms.
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
Calc. |
|
Expt. [2] |
|
| |
|
|
|
|
|
|
|
|
D(5) |
Xaa(35Cl) |
- |
67.35
|
- |
68.08(16)
|
|
|
|
Xbb |
|
19.33
|
|
18.32(16)
|
|
|
|
Xaa(14N) |
|
4.87
|
|
4.64(20)
|
|
|
|
Xbb |
|
- 0.05
|
|
- 0.08(12)
|
|
|
|
|
|
|
|
|
|
|
D(6)
|
Xaa(35Cl) |
-
|
64.46
|
-
|
64.91(16)
|
|
|
|
Xbb
|
|
16.60
|
|
14.78(15)
|
|
|
|
Xaa(14N) |
|
4.78
|
|
4.53(19)
|
|
|
|
Xbb |
|
0.50
|
|
0.25(11)
|
|
|
|
|
|
|
|
|
|
|
D(7) |
Xaa(35Cl) |
-
|
64.11
|
-
|
64.78(11) |
|
|
|
Xbb |
|
16.09
|
|
15.78(12)
|
|
|
|
Xaa(14N) |
|
4.76
|
|
4.42(18)
|
|
|
|
Xbb |
|
- 0.49
|
|
- 1.01(12)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
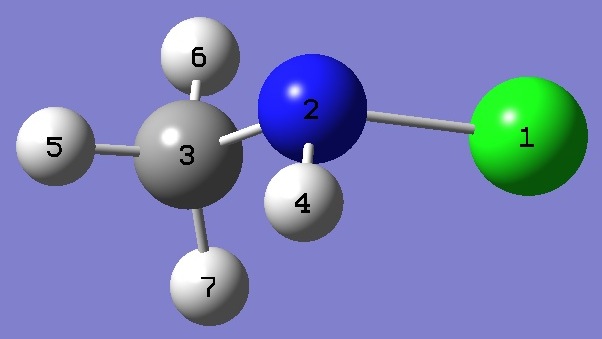
| Table 3.
CH3NHCl Selected structure parameters, ropt and ro [2]
(Å and degrees). The complete ropt structure is given here in Z-Matrix format. |
| |
|
|
|
|
|
ropt |
ro |
|
|
|
| ClN |
1.7475 |
1.751(3) |
| NC |
1.4639 |
1.467(6) |
| ClNC |
108.88 |
109.32(17) |
| NH |
1.0167 |
1.012(2) |
| Click on image to enlarge. |
ClNH |
102.69 |
102.23(17) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
| Table 4. CH3NHCl Rotational constants (MHz). Normal species. |
| |
|
|
|
|
|
Calc. ropt |
Expt. [1] |
|
|
|
|
|
A |
36 258.0 |
36 204.2(6) |
|
B |
5 992.0 |
5 935.8(2) |
|
C |
5 391.9 |
5 340.4(2) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
[1] A.M.Mirri and W.Caminati, J.Mol.Spectrosc. 47,204(1973). |
|
|
[2] W.Caminati, R.Cervellati, and A.M.Mirri J.Mol.Spectrosc. 51,288(1974).
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH3NH2 |
NH2Cl |
(CH3)2NH |
NF2H |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table of Contents |
|
|
|
|
|
Molecules/Chlorine |
|
|
|
|
|
Molecules/Nitrogen |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH3NHCl.html |
|
|
|
|
|
|
Last
Modified 12 May 2008 |
|
|
|
|
|
|
|
|
|
|

