|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
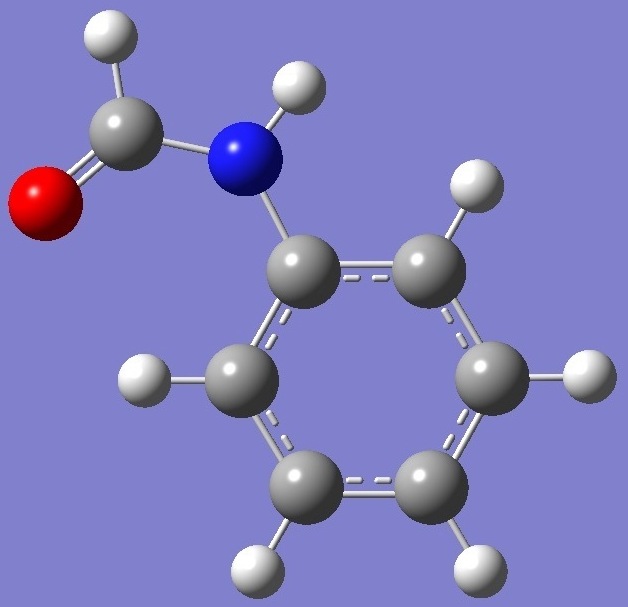
HC(=O)-N(H)-C6H5
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Nitrogen |
|
|
|
Nuclear
Quadrupole Coupling Constants |
|
|
in
trans-Formanilide
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Calculation of the
nitrogen nqcc's in formanilide was made here on molecular
structures given by HF/6-311+G(d,p) and HF/6-311++G(3df,3pd)
optimizations, with Cs symmetry assumed. These
are
compared with the
experimental nqcc's [1] in Table 1. Structure parameters are
given in Table 2, rotational constants in Table 3.
|
|
|
|
|
|
|
|
|
|
|
|
|
In Table 1, subscripts a,b,c refer to
the
principal axes of the inertia tensor; subscripts x,y,z to the principal
axes
of the nqcc tensor. ETA = (Xxx - Xyy)/Xzz. |
|
|
RMS is the root
mean square difference between calculated and experimental diagonal
nqcc's (percentage of the average of the magnitudes of the experimental
nqcc's). RSD is the calibration residual standard deviation of
the B3PW91/6-311+G(df,pd) model for calculation of the nitrogen nqcc's.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
Table 1. 14N nqcc's in Formanilide
(MHz). Calculation was made on the (1) HF/6-311+G(d,p) and (2)
HF/6-311++G(3df,3pd) optimized structures. |
|
| |
|
|
|
|
|
|
|
|
|
|
|
Calc. (1)
|
|
Calc. (2) |
|
Expt. [1] |
|
| |
|
|
|
|
|
|
|
|
|
Xaa |
|
1.863 |
|
1.843 |
|
1.926(14) |
|
|
Xbb |
|
1.718 |
|
1.709 |
|
1.745(17) |
|
|
Xcc |
- |
3.581 |
- |
3.552 |
- |
3.671 |
|
|
|Xab| |
|
0.252 |
|
0.254 |
|
|
|
|
|
|
|
|
|
|
|
|
|
RMS |
|
0.065 (2.7 %) |
|
0.086 (3.5 %) |
|
|
|
|
RSD |
|
0.030 (1.3 %) |
0.030 (1.3 %) |
|
|
|
|
|
|
|
|
|
|
|
|
Xxx |
|
1.528 |
|
1.514 |
|
|
|
|
Xyy |
|
2.052 |
|
2.039 |
|
|
|
|
Xzz |
- |
3.581 |
- |
3.552 |
|
|
|
|
ETA |
|
0.146 |
|
0.148 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
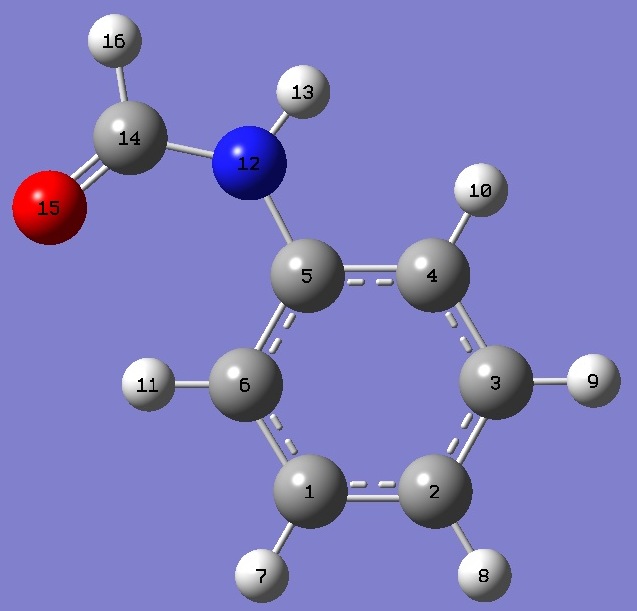
| Table 2. Formanilide.
Heavy atom structure parameters (Å and
degrees). Complete structures are given here in Z-Matrix format. |
| |
|
|
|
|
ropt (1) =
HF/6-311+G(d,p) optimization. |
|
ropt (2) =
HF/6-311++G(3df,3pd) optimization. |
| |
|
|
|
| Point Group: Cs |
|
ropt (1) |
ropt (2) |
|
|
|
|
|
C(5)N |
1.4102 |
1.4063 |
| NH(13) |
0.9934 |
0.9912 |
| NC(14) |
1.3562 |
1.3526 |
| C(14)H(16) |
1.0923 |
1.0908 |
| C(14)O |
1.1878 |
1.1855 |
| C(5)NH(13) |
115.53 |
115.50 |
| C(5)NC(14) |
129.31 |
129.46 |
| NC(14)H(16) |
111.08 |
111.08 |
| NC(14)O |
127.10 |
127.11 |
|
|
|
| C(5)C(6) |
1.3892 |
1.3861 |
| C(6)C(1) |
1.3868 |
1.3833 |
| C(1)C(2) |
1.3831 |
1.3797 |
| C(2)C(3) |
1.3858 |
1.3823 |
| C(3)C(4) |
1.3812 |
1.3776 |
| C(4)C(5) |
1.3919 |
1.3887 |
| C(4)C(5)C(6) |
119.55 |
119.52 |
|
C(5)C(6)C(1) |
119.25 |
119.25 |
|
C(6)C(1)C(2) |
121.40 |
121.45 |
|
C(1)C(2)C(3) |
119.00 |
118.95 |
|
C(2)C(3)C(4) |
120.31 |
120.34 |
|
C(3)C(4)C(5) |
120.48 |
120.50 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
| Table 3. Formanilide.
Rotational Constants (MHz). |
| |
|
|
|
|
|
ropt (1) =
HF/6-311+G(d,p) optimization. |
|
ropt (2)
= HF/6-311++G(3df,3pd) optimization. |
| |
|
|
|
|
|
|
Calc. ropt (1) |
Calc. ropt (2) |
Expt. [1] |
|
|
|
|
|
|
A |
4319.5 |
4343.5 |
4214.4567(14) |
|
B |
1127.0 |
1131.2 |
1123.6904(4) |
|
C |
893.8 |
897.5 |
888.0883(4) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
[1] J.-R.Aviles Moreno, T.R.Huet,
D.Petitprez, J.Mol.Struct. 780-781,234(2006).
|
|
|
|
|
|
|
|
|
|
|
|
|
S.Blanco, J.C.López,
A.Lesarri, W.Caminati, and J.L.Alonso, Mol.Phys. 103,1473(2005):
(3/2)Xaa = 2.890(6) and (1/4)(Xbb - Xcc)
= 1.354(2) MHz ==> Xaa = 1.926(4), Xbb =
1.745, and Xcc = -3.671 MHz which, aside from the
uncertainties, are the same as that given in Table 1. |
|
|
|
|
|
|
|
|
|
|
|
|
P.Ottaviani, S.Melandri, A.Maris, P.G.Favero, W.Caminati, J.Mol.Spectrosc. 205,173(2001): (Xbb - Xcc)
= 5.45(6) MHz.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Formamide |
Acetamide |
N-Ethylformamide |
|
|
|
N-Acetylglycine |
Dimethylamine |
cis-Formanilide |
|
|
N-Methylacetamide |
Propionamide |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table of Contents |
|
|
|
|
|
Molecules/Nitrogen |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Formanilide.html |
|
|
|
|
|
|
Last
Modified 10 Jan 2011 |
|
|
|
|
|
|
|
|
|
|

