|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH3-CHCl-CH2Cl |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Chlorine |
|
|
|
Nuclear
Quadrupole Coupling Constants |
|
|
in
1,2-Dichloropropane
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Calculation was made of the complete
nqcc tensors for 35Cl
and 37Cl
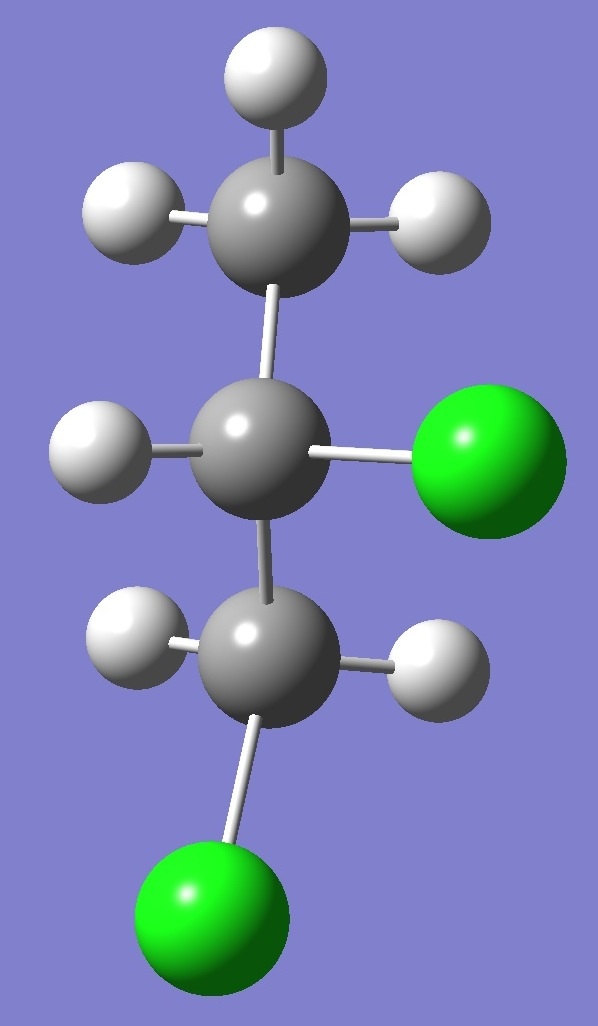
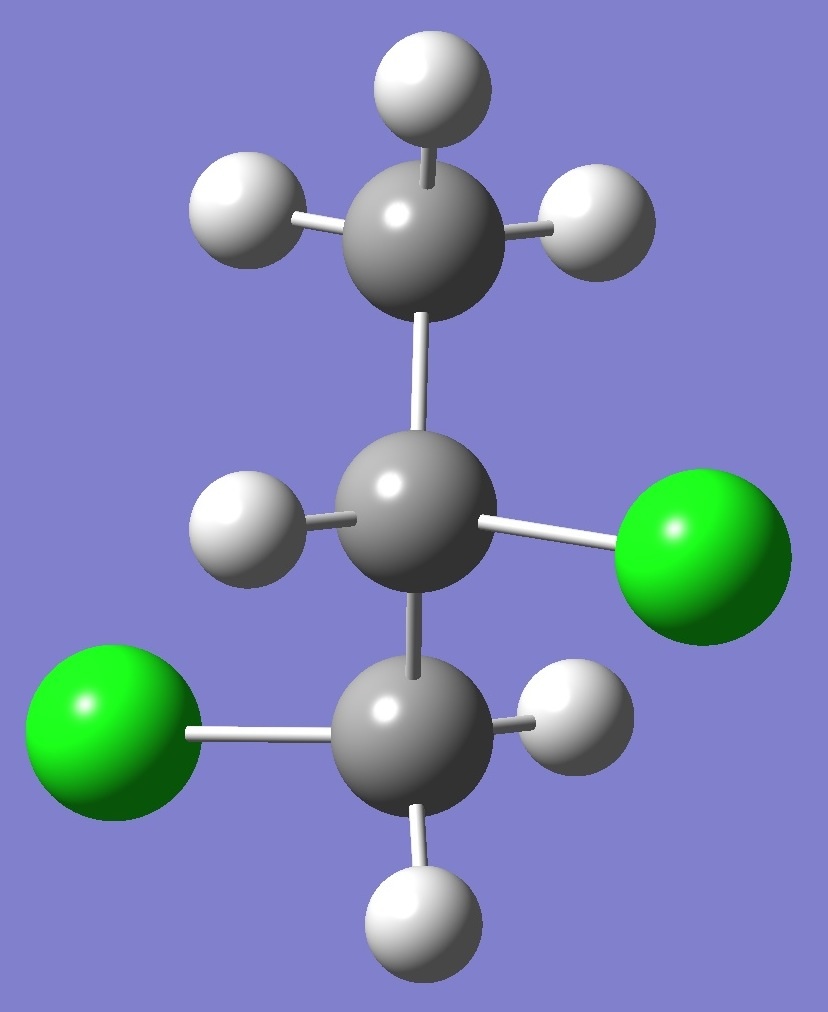
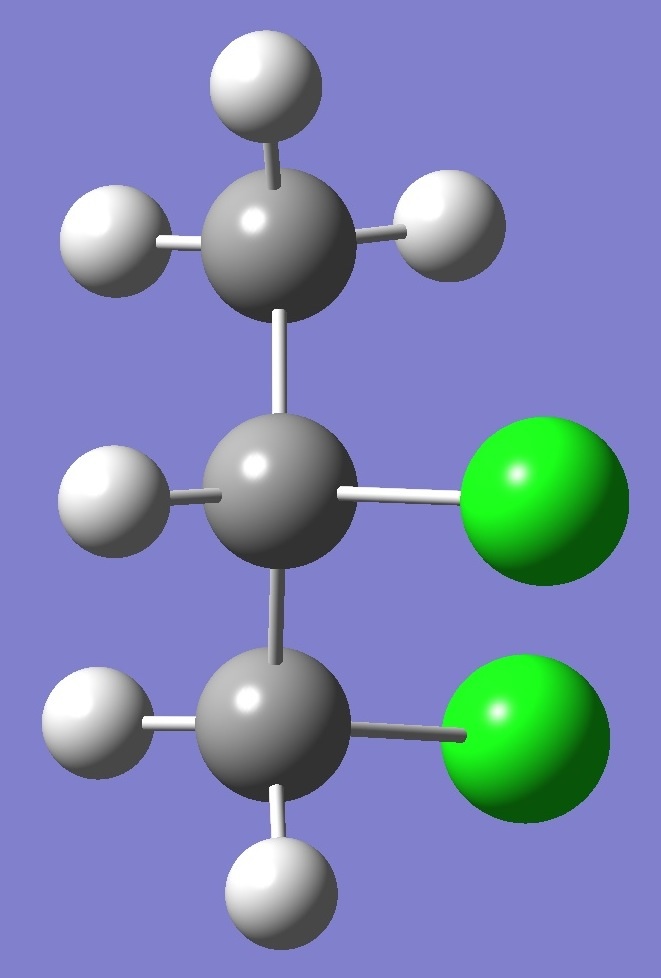
in each of three structural conformers of 1,2-dichloropropane shown
below: |
|
|
|
|
|
|
|
|
|
|
|
|
G- |
|
A |
|
G+ |
|
|
|
|
|
|
|
|
|
|
|
|
 |
|
|
|
 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Results for conformer G+
are given on
this page. To see the results for conformers A and
G- click on the corresponding image.
|
|
|
|
|
|
|
|
|
|
|
|
|
Calculation was made
on MP2/6-311+G(3df,3pd) and MP2/aug-cc-pVTZ optimized structures, each
with approximate equilibrium bond lengths. Calculated nqcc
tensors are given below in Tables 1 - 5, structure parameters in
Table 6. Rotational constants and electric dipole moments are
given in Table 7. |
|
|
|
|
|
|
|
|
|
|
|
|
Energies relative to conformer A are,
for the MP2/6-311+G(3df,3pd) optimized structures, G+ =
5.50 and G- = 6.07 kJ/mole; and for
the MP2/aug-cc-pVTZ optimized structures, G+ =
5.96 and G- = 6.13 kJ/mole. |
|
|
|
|
|
|
|
|
|
|
|
|
In Tables 1 - 5, subscripts a,b,c
refer to the
principal axes of the inertia tensor; x,y,z to the principal axes
of the nqcc tensor.
Øz,CCl (degrees) is the angle between the principal
z-axis of the
nqcc tensor and the CCl bond axis. ETA = (Xxx - Xyy)/Xzz. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
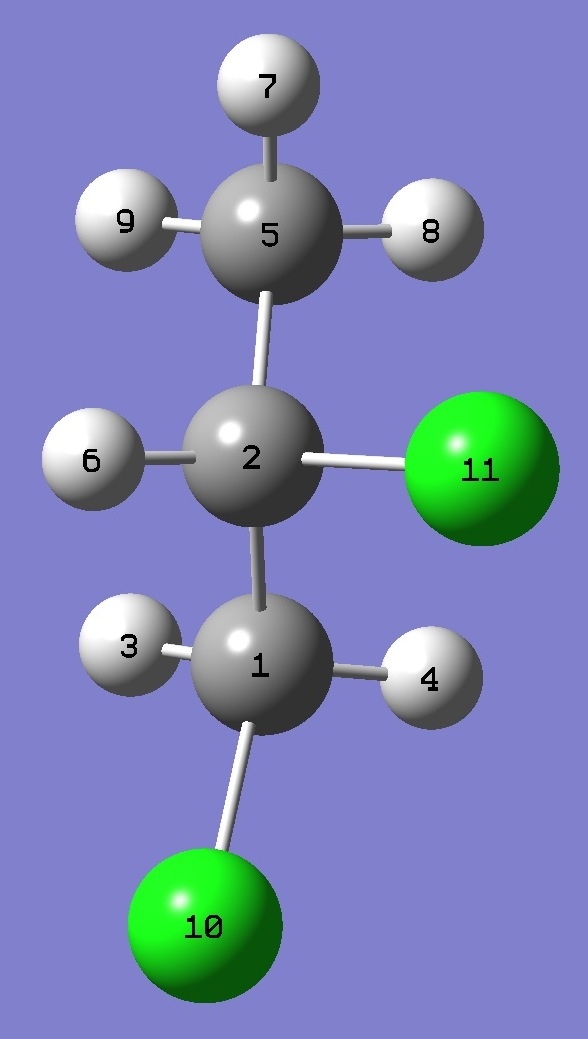
Table 1.
35Cl(10)
nqcc's in Conformer G+ of CH3-CH35Cl-CH235Cl(10)
(MHz). Calculation was made on the (1) MP2/6-311+G(3df,3pd)
and (2) MP2/aug-cc-pVTZ optimized structures, each with approximate
equilibrium bond lengths. See below for atomic numbering. |
|
| |
|
|
|
|
|
|
|
|
| |
|
|
Calc (1) |
|
Calc (2) |
|
Expt.
|
|
| |
|
|
|
|
|
|
|
|
|
Xaa |
|
- 4.72 |
|
- 4.62 |
|
|
|
| |
Xbb |
|
26.64 |
|
26.81 |
|
|
|
|
Xcc |
- |
21.92 |
- |
22.19 |
|
|
|
|
Xab |
- |
21.88 |
- |
21.69 |
|
|
|
|
Xac |
- |
48.32 |
- |
48.36 |
|
|
|
|
Xbc |
- |
25.07 |
- |
24.90 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Xxx |
|
35.56 |
|
35.54 |
|
|
|
|
Xyy |
|
37.89 |
|
37.89 |
|
|
|
| |
Xzz |
- |
73.46 |
- |
73.43 |
|
|
|
|
ETA |
|
0.0317 |
|
0.0320 |
|
|
|
|
Øz,CCl |
|
0.35 |
|
0.37 |
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
Table 2.
35Cl(11)
nqcc's in Conformer G+ of CH3-CH35Cl(11)-CH237Cl
(MHz). Calculation was made on the (1) MP2/6-311+G(3df,3pd)
and (2) MP2/aug-cc-pVTZ optimized structures, each with approximate
equilibrium bond lengths. See below for atomic numbering. |
|
| |
|
|
|
|
|
|
|
|
| |
|
|
Calc (1) |
|
Calc (2) |
|
Expt.
|
|
| |
|
|
|
|
|
|
|
|
|
Xaa |
|
8.11 |
|
8.21 |
|
|
|
| |
Xbb |
- |
30.30 |
- |
30.30 |
|
|
|
|
Xcc |
|
22.19 |
|
22.08 |
|
|
|
|
Xab |
|
42.64 |
|
42.54 |
|
|
|
|
Xac |
|
19.64 |
|
19.66 |
|
|
|
|
Xbc |
- |
29.77 |
- |
22.08 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Xxx |
|
35.40 |
|
35.37 |
|
|
|
|
Xyy |
|
36.04 |
|
36.03 |
|
|
|
| |
Xzz |
- |
71.44 |
- |
71.40 |
|
|
|
|
ETA |
|
0.0088 |
|
0.0091 |
|
|
|
|
Øz,CCl |
|
0.28 |
|
0.28 |
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
Table 3.
Cl
nqcc's in Conformer G+ of CH3-CH37Cl(11)-CH235Cl(10)
(MHz). Calculation was made on the (1) MP2/6-311+G(3df,3pd)
and (2)
MP2/aug-cc-pVTZ optimized structures, each with approximate equilibrium
bond lengths. See below for atomic numbering. |
|
| |
|
|
|
|
|
|
|
|
| |
35Cl(10) |
|
Calc (1) |
|
Calc (2) |
|
Expt.
|
|
| |
|
|
|
|
|
|
|
|
|
Xaa |
|
- 3.79 |
|
- 3.70 |
|
|
|
| |
Xbb |
|
25.72 |
|
25.90 |
|
|
|
|
Xcc |
- |
21.93 |
- |
22.20 |
|
|
|
|
Xab |
- |
22.51 |
- |
22.32 |
|
|
|
|
Xac |
- |
47.78 |
- |
47.82 |
|
|
|
|
Xbc |
- |
26.08 |
- |
25.92 |
|
|
|
|
|
|
|
|
|
|
|
|
|
37Cl(11) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Xaa |
|
5.20 |
|
5.28 |
|
|
|
| |
Xbb |
- |
22.90 |
- |
22.91 |
|
|
|
|
Xcc |
|
17.71 |
|
17.63 |
|
|
|
|
Xab |
|
34.19 |
|
34.12 |
|
|
|
|
Xac |
|
15.73 |
|
15.74 |
|
|
|
|
Xbc |
- |
23.00 |
- |
23.05 |
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
Table 4.
Cl
nqcc's in Conformer G+ of CH3-CH35Cl(11)-CH237Cl(10)
(MHz). Calculation was made on the (1) MP2/6-311+G(3df,3pd)
and (2)
MP2/aug-cc-pVTZ optimized structures, each with approximate equilibrium
bond lengths. See below for atomic numbering. |
|
| |
|
|
|
|
|
|
|
|
| |
35Cl(11) |
|
Calc (1) |
|
Calc (2) |
|
Expt.
|
|
| |
|
|
|
|
|
|
|
|
|
Xaa |
|
9.04 |
|
9.15 |
|
|
|
| |
Xbb |
- |
31.25 |
- |
31.25 |
|
|
|
|
Xcc |
|
22.21 |
|
22.10 |
|
|
|
|
Xab |
|
42.21 |
|
42.12 |
|
|
|
|
Xac |
|
19.29 |
|
19.31 |
|
|
|
|
Xbc |
- |
29.96 |
- |
30.03 |
|
|
|
|
|
|
|
|
|
|
|
|
|
37Cl(10) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Xaa |
|
- 4.41 |
|
- 4.33 |
|
|
|
| |
Xbb |
|
21.15 |
|
21.28 |
|
|
|
|
Xcc |
- |
16.74 |
- |
16.94 |
|
|
|
|
Xab |
- |
17.27 |
- |
17.12 |
|
|
|
|
Xac |
- |
38.25 |
- |
38.28 |
|
|
|
|
Xbc |
- |
19.48 |
- |
19.35 |
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
Table 5.
37Cl
nqcc's in Conformer G+ of CH3-CH37Cl(11)-CH237Cl(10)
(MHz). Calculation was made on the (1) MP2/6-311+G(3df,3pd)
and (2)
MP2/aug-cc-pVTZ optimized structures, each with approximate equilibrium
bond lengths. See below for atomic numbering. |
|
| |
|
|
|
|
|
|
|
|
| |
37Cl(11) |
|
Calc (1) |
|
Calc (2) |
|
Expt.
|
|
| |
|
|
|
|
|
|
|
|
|
Xaa |
|
5.96 |
|
6.05 |
|
|
|
| |
Xbb |
- |
23.69 |
- |
23.69 |
|
|
|
|
Xcc |
|
17.72 |
|
17.65 |
|
|
|
|
Xab |
|
33.87 |
|
33.80 |
|
|
|
|
Xac |
|
14.45 |
|
15.47 |
|
|
|
|
Xbc |
- |
23.15 |
- |
23.20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
37Cl(10) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Xaa |
|
- 3.69 |
|
- 3.61 |
|
|
|
| |
Xbb |
|
20.43 |
|
20.56 |
|
|
|
|
Xcc |
- |
16.74 |
- |
16.95 |
|
|
|
|
Xab |
- |
17.78 |
- |
17.63 |
|
|
|
|
Xac |
- |
37.84 |
- |
37.87 |
|
|
|
|
Xbc |
- |
20.27 |
- |
20.14 |
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Table 6. CH3-CHCl-CH2Cl,
conformer G+
Heavy atom structure parameters (Å and
degrees). r(1) = MP2/6-311+G(3df,3pd) and r(2) = MP2/aug-cc-pVTZ
optimized structures, each with approximate equilibrium bond lengths.
Complete structures are given here
in Z-matrix format. |
|
|
|
|
|
|
|
|
r(1) |
r(2) |
|
 |
|
|
|
|
Cl(10)C(1) |
1.7770 |
1.7781 |
|
C(1)C(2) |
1.5138 |
1.5131 |
|
C(2)Cl(11) |
1.7884 |
1.7896 |
|
C(2)C(5) |
1.5117 |
1.5114 |
|
C(1)C(2)C(5) |
113.93 |
114.15 |
|
C(2)C(1)C1(10) |
113.08 |
112.91 |
|
C(1)C(2)Cl(11) |
110.12 |
109.91 |
|
ClC(1)C(2)Cl |
- 67.22 |
-67.39 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
| Table 7. CH3-CH35Cl-CH235Cl,
conformer G+ Rotational
Constants (MHz), and Dipole Moments
(D) on the (1) MP2/6-311+G(3df,3pd) and (2) MP2/aug-cc-pVTZ
optimized structures, each with approximate equilibrium bond lengths. |
| |
|
|
|
|
| |
|
Calc (1) |
Calc (2) |
Expt. |
|
|
|
|
|
|
A |
4436.34 |
4435.27 |
|
|
B |
2026.95 |
2031.00 |
|
|
C |
1690.11 |
1694.26 |
|
|
|
|
|
|
|
|µa| |
0.17 |
0.17 |
|
|
|µb| |
2.09 |
2.09 |
|
|
|µc| |
1.67 |
1.68 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
t-1-Chloropropane |
g-1-Chloropropane |
2-Chloropropane |
2,2-Dichloropropane |
|
|
2,2-Chlorofluoropropane |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
G.A.Guirgis, Y.D.Hsu, A.C.Vlaservich,
H.D.Stidham, and J.R.Durig, J.Mol.Struct. 378,83(1996). IR and
Raman. |
|
|
S.H.Schei and R.Stølevik,
J.Mol.Struct. 128,171(1985). ged |
|
|
|
|
|
|
|
|
|
|
|
|
Table of Contents |
|
|
|
|
|
Molecules/Chlorine |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CH3CHClCH2Cl_G_plus.html |
|
|
|
|
|
|
Last
Modified 22 Oct 2010 |
|
|
|
|
|
|
|
|
|
|



